For centuries, losing a tooth was a permanent problem. Whether it was a childhood sports injury or a cavity that went too far, once your adult teeth were gone, that was it. You were left with two choices: living with the gap or opting for a metal-and-ceramic substitute.
But as of 2026, we are entering a new era. We are no longer just talking about better prosthetics; we are talking about biological restoration. The world’s first "teething medicine"—a drug capable of triggering the body to regrow its own teeth—has moved from the realm of science fiction into active human clinical trials.
The Science: Waking Up Our "Third Set"
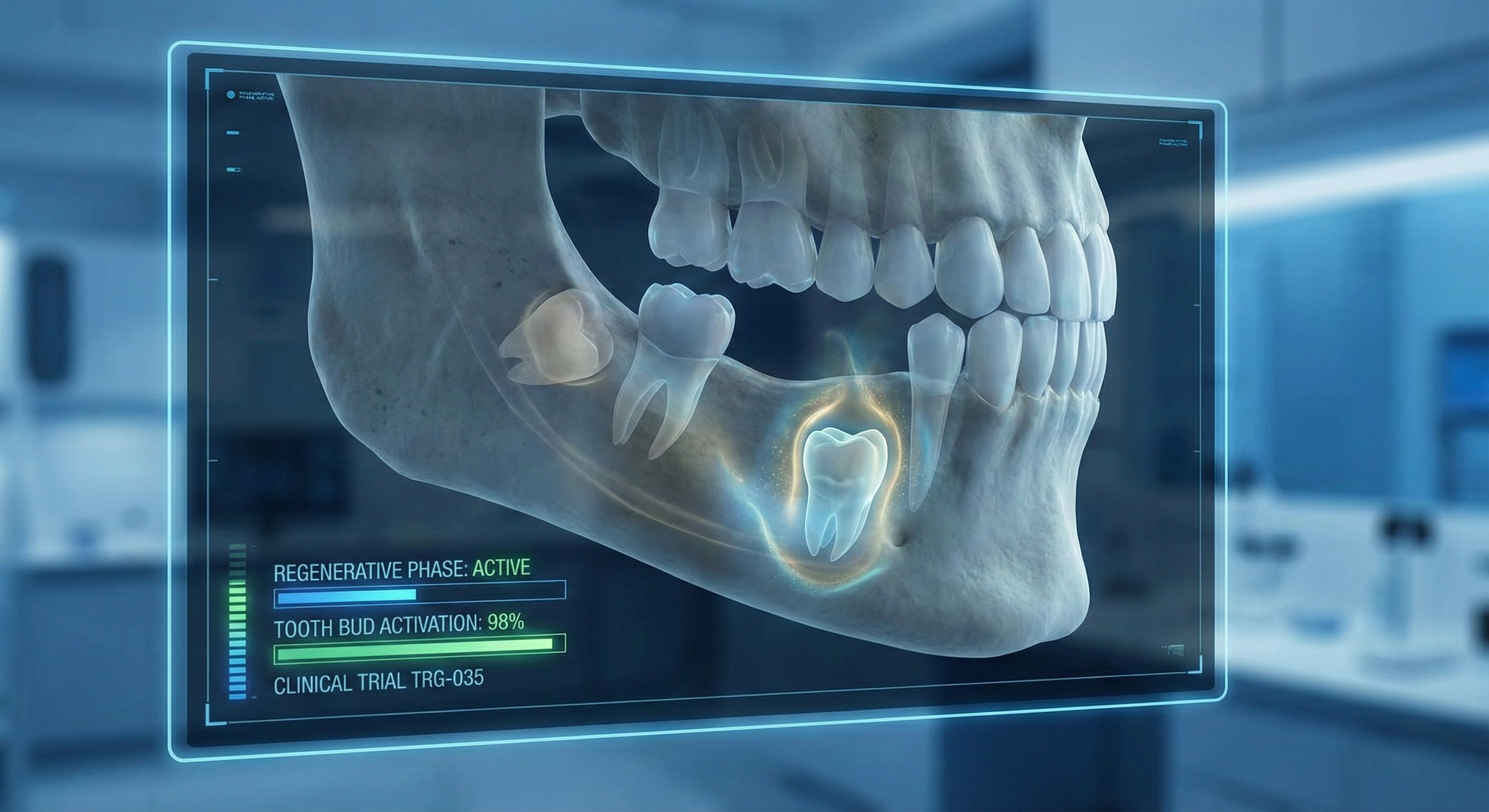
The logic behind this breakthrough is fascinatingly simple. Human beings actually carry the "seeds" for a third set of teeth deep within our gums. However, a specific protein called USAG-1 acts like a biological brake, keeping these buds dormant for our entire adult lives.
Researchers at Kyoto University and the startup Toregem Biopharma developed a monoclonal antibody called TRG035. This drug essentially "cuts the brake lines." By neutralizing the USAG-1 protein, it allows those dormant tooth buds to wake up and develop into full, functional teeth—naturally.
The Road to Reality: 2024 to 2026
The transition from laboratory success to human healing has been moving at an impressive pace:
- The Foundation: Initial studies in Science Advances showed the drug worked perfectly in mice and ferrets, which share similar dental "blueprints" with humans.
- Phase 1 Safety (2024-2025): Clinical trials began at Kyoto University Hospital with 30 healthy adult men. The focus was simple: proving the drug was safe for human use.
- Current Focus (2026): Trials have now expanded to children aged 2 to 7 who suffer from congenital anodontia (born without some or all teeth). This is the "frontier group" that could see the first life-changing results.
The Move to America
While the research began in Japan, the United States is now firmly in the loop. In late November 2025, Toregem Biopharma reached a major regulatory milestone: a formal response from the U.S. Food and Drug Administration (FDA) following a Pre-IND meeting.
This essentially gives the team a green light and a roadmap for U.S.-based clinical trials, which are expected to begin by late 2026. Thanks to recent FDA policy shifts designed to fast-track breakthrough therapies, American patients might have access to this "teething medicine" sooner than originally anticipated.
What the Future Holds
The ultimate goal is commercial availability by 2030. If the trials continue their current success, your future visit to the dentist might look very different. Instead of a surgical procedure involving titanium screws and bone grafts, you might receive a series of intravenous treatments that trigger your body to grow a real, enamel-covered tooth.
"We want to offer a third choice," says Dr. Katsu Takahashi, the lead researcher. "One that sits alongside dentures and implants but offers the durability and feel of the real thing." At a projected cost of roughly $10,000, it’s being positioned to compete with high-end implants while offering a result that is entirely your own.
Sources & Further Reading
- Scientific Basis: “Anti–USAG-1 therapy for tooth regeneration through enhanced BMP signaling,” Science Advances (2021).
- Clinical Trial Update: “Japanese startup developing world's first drug to grow new teeth,” The Japan Times (2024-2026).
- Regulatory Milestone: “Notification of receipt of response regarding Pre-IND meeting with the US FDA,” Toregem BioPharma Official Press Release (Nov 27, 2025).
- ADA Recognition: “Can teeth be regrown?” American Dental Association (ADA) News.
-1.png?width=1182&height=222&name=TSS_alternate_logo_v1b%20(long)-1.png)

.png?width=160&height=90&name=cdn%20(1).png)